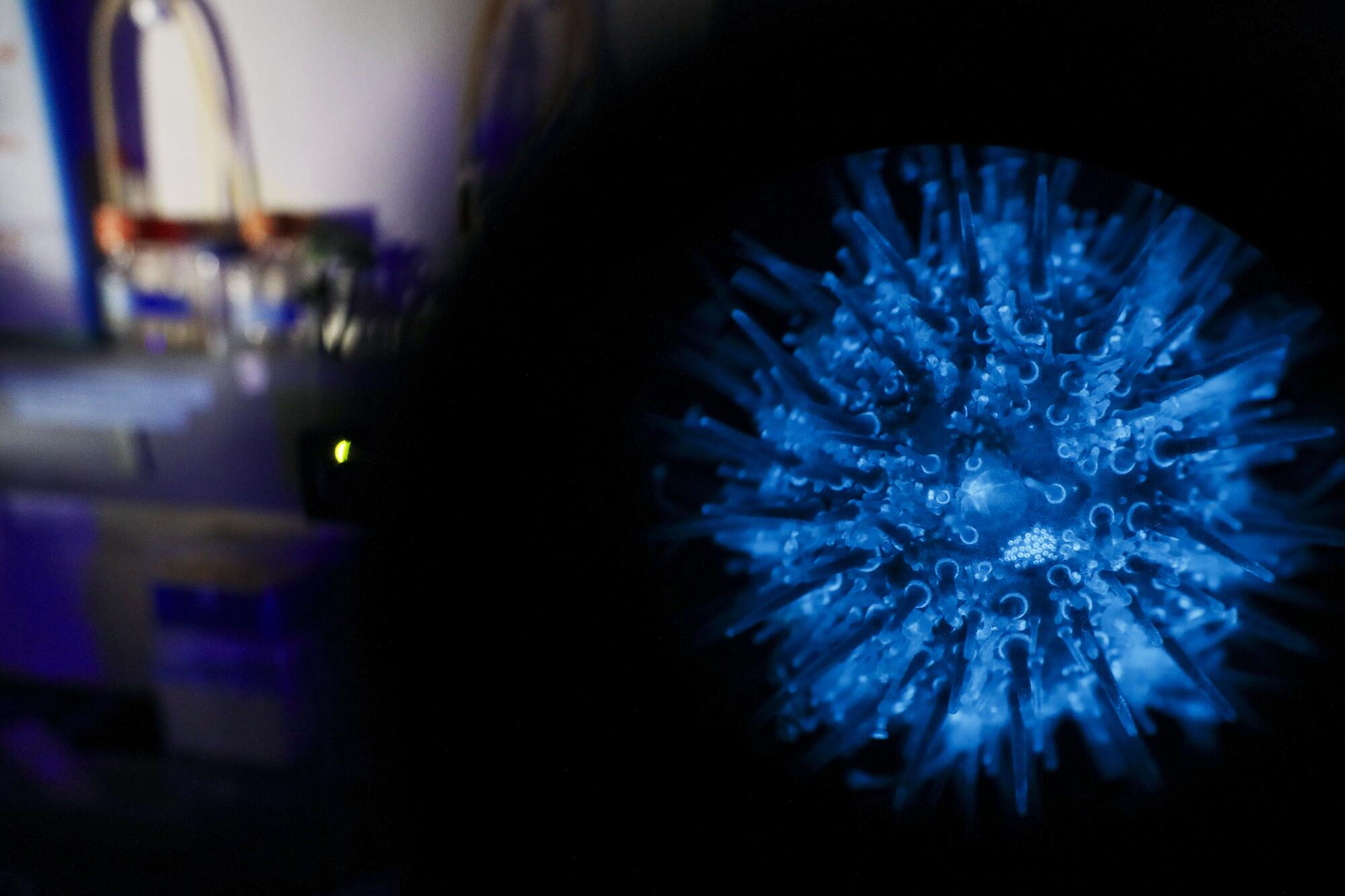
Consider the sea urchin. Specifically, the painted urchin: Lytechinus pictus, a prickly Ping-Pong ball from the eastern Pacific Ocean.
The species is a smaller and shorter-spined cousin of the purple urchins devouring kelp forests. They produce massive numbers of sperm and eggs that fertilize outside of their bodies, allowing scientists to watch the process of urchin creation up close and at scale. One generation gives rise to the next in four to six months. They share more genetic material with humans than fruit flies do and can’t fly away — in short, an ideal lab animal for the developmental biologist.
Scientists have been using sea urchins to study cell development for roughly 150 years. Despite urchins’ status as super reproducers, practical concerns often compel scientists to focus their work on more easily accessible animals: mice, fruit flies, worms.
Scientists working with mice, for example, can order animals online with the specific genetic properties they are hoping to study — transgenic animals, whose genes have been artificially tinkered with to express or repress certain traits.
Researchers working with urchins typically have to spend part of their year collecting them from the ocean.
“Can you imagine if mouse researchers were setting a mousetrap every night, and whatever it is they caught is what they studied?” said Amro Hamdoun, a professor at UC San Diego’s Scripps Institution of Oceanography.

Marine invertebrates represent about 40% of the animal world’s biological diversity yet appear in a scant fraction of a percentage of animal-based studies. What if researchers could access sea urchins as easily as mice? What if it were possible to make and raise lines of transgenic urchins?
How much more could we learn about how life works?
“You know how during the pandemic, everyone was making sourdough? I’m not good at making sourdough,” Hamdoun said recently at his office in Scripps’ Hubbs Hall. He set his sights instead on a project of a different sort: a new transgenic lab animal, “a fruit fly from the sea.”
In March, Hamdoun’s lab published a paper on the bioRxiv preprint server demonstrating the successful insertion of a piece of foreign DNA — specifically, a fluorescent protein from a jellyfish — into the genome of a painted urchin that passed the change down to its offspring.
The result is the first transgenic sea urchin, one that happens to glow like a Christmas bulb under a fluorescent light. (The paper has been submitted for peer review.)
The animals are the first transgenic echinoderms, the phylum that includes starfish, sea cucumbers and other marine animals. Hamdoun’s mission is to make genetically modified urchins available to researchers anywhere, not just those who happen to work in research facilities at the edge of the Pacific Ocean.

“If you look at some of the other model organisms, like Drosophila [fruit flies], zebrafish and mouse, there are well-established resource centers,” said Elliot Jackson, a postdoctoral researcher at Scripps and lead author of the paper. “If you want a transgenic line that labels the nervous system, you could probably get that. You could order it. And that’s what we hope we can be for sea urchins.”
Being able to genetically modify an animal supercharges what scientists can learn from it, with implications far beyond any individual species.
“It will transform sea urchins as a model for understanding neurobiology, for understanding developmental biology, for understanding toxicology,” said Christopher Lowe, a Stanford professor of biology who was not involved in the research.
What are those blue things washing up on Southern California beaches? Velella velella, of course. Also known as by-the-wind sailors. They’re kind of like jellyfish.
The lab’s breakthrough, and its focus on making the animals freely available to fellow scientists, will “allow us to explore how evolution has solved a lot of really complicated life problems,” he said.
Researchers tend to study mice, flies and the like not because the animals’ biology is best suited to answer their questions but because “all the tools that were necessary to get at your questions were built up in just a few species,” said Deirdre Lyons, an associate professor of biology at Scripps who worked with Hamdoun on early research related to the project.
Expanding the range of animals available for sophisticated lab work is like adding colors to an artist’s palette, Lyons said: “Now you can go get the color that you really want, that best fits your vision, rather than being stuck with a few models.”

On the ground floor of Hamdoun’s office building is the Hubbs Hall experimental aquarium, a garage-like space crammed with tanks full of recirculating seawater and a motley assortment of marine life.
On a recent visit, Hamdoun reached into a tank and gently dislodged a painted urchin. It scooched with surprising speed across an outstretched palm, as if exploring alien terrain.
The last common ancestor of L. pictus and Homo sapiens lived at least 550 million years ago. Despite the different evolutionary paths we’ve since traveled, our genomes reveal a shared biological heritage.
The genetic instructions that drive the transformation of a single zygote into a living body are strikingly similar in our two species. Specialized systems differentiating from a single fertilized egg and the translation of a jumble of proteins into a singular living thing — on the cellular level, all of that proceeds in much the same way for urchins and people.
These animals are “really fundamental to our understanding of all of life,” Hamdoun said, placing the urchin back in its tank. “And historically, very inaccessible genetically.”
The experimental aquarium was built in the 1970s, when scooping life from the sea was the only way to acquire research specimens. A few floors up in Hubbs Hall, Hamdoun led the way into the urchin nursery — the first large-scale effort to raise successive generations of the animals in a laboratory. At any given moment, the team has 1,000 to 2,000 sea urchins in various stages of development.

Row upon row of tiny plastic tanks stood against a wall, each containing a lentil-size juvenile urchin. A strip of tape on each tank noted the animal’s genetic modification and date of fertilization. On some, a second bit of tape indicated animals that had the modification in their sex cells’ DNA, meaning it could be passed down to offspring. (For this reason, the lab keeps its urchins scrupulously separate from the wild population.)
“One of the big questions in all of biology is to understand how the series of instructions in the genome gives you whatever phenotype you want to study,” Hamdoun said — essentially, how the string of amino acids that is an animal’s genetic code gives rise to the characteristics of the living, respiring creature. “One of the fundamental things you have to do is be able to modify that genome, and then study what the outcome is.”
The pup could become Aquarium of the Pacific’s first surrogate-raised otter to return to the wild — if she masters the skills needed to hack it in the ocean.
He pointed to a tank containing a tiny urchin from whose genetic code the protein ABCD1 has been snipped.
ABCD1 acts like a bouncer, Hamdoun explained, parking along the cell membrane and ejecting foreign molecules. The protein’s action can preserve the cell from harmful substances but can sometimes work against an organism’s best interest, as when it prevents the cell from absorbing a necessary medication.
Researchers using urchins in which that protein no longer works can study the movement of a molecule through an organism — DDT, for example — and measure how much of the substance ends up in the cell without the confounding interference of ABCD1. They can reverse-engineer how big a role ABCD1 plays in preventing a cell from absorbing a drug.
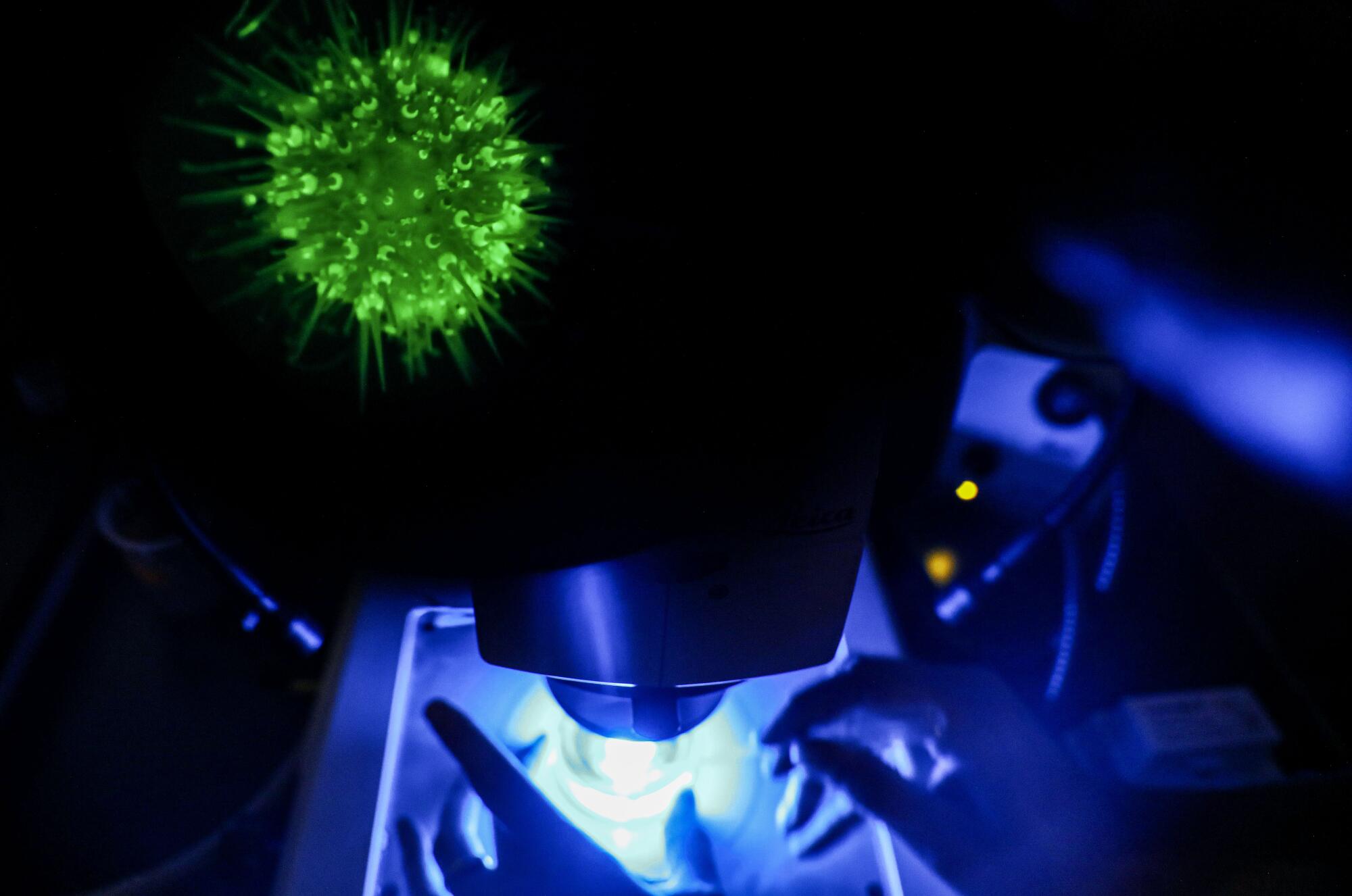
And then there are the fluorescent urchins.
“The magic happens in this room,” Jackson said, walking into a narrow office with $1 million worth of microscopes at one end and a decades-old hand-cranked centrifuge bolted to a table at another.
He placed a petri dish containing three pencil-eraser-size transgenic urchins under a microscope. At 120 times its size, each looked like the Times Square New Year’s Eve ball come to life — a glowing, wiggling creature of pentamerous radial symmetry.
Fluorescence is not just an echinoderm party trick. Lighting up the cells makes it easier for researchers to track their movement in a developing organism. Researchers can watch as the early cells of a blastula divide and reorganize into neural or cardiac tissue. Eventually, scientists will be able to turn off individual genes and see how that affects development. It will help us understand how our own species develops, and why that development doesn’t always proceed according to plan.
USC’s Berislav Zlokovic has faced questions about the integrity of his research. Since a whistleblower report last year, several papers have been retracted and a drug trial has been paused.
The lab has “done a great job. It’s really been welcomed by the community,” said Marko Horb, senior scientist and director of the National Xenopus Resource at the University of Chicago’s Marine Biological Laboratory.
Horb runs the national clearinghouse for genetically modified species of Xenopus, a clawed frog used in lab research. Funded in part by the National Institutes of Health, the center develops lines of transgenic frogs for scientific use and distributes them to researchers.
Hamdoun envisions a similar resource center for his lab’s urchins. They’ve already started sending tiny vials of transgenic urchin sperm to interested scientists, who can grow bespoke urchins with eggs acquired from Hamdoun’s lab or another source.
Hamdoun vividly recalls the time he spent earlier in his career trying to track down random snippets of DNA necessary for his research, the disappointment and frustration of writing to professors and former postdocs only to find that the material had long been lost. He’d rather future generations of scientists spend their time on discovery.
“Biology is really interesting,” he said. “The more people can get access to it, the more we’re going to learn.”











